Current aspects and prospects of glass ionomer cements for clinical dentistry
Article information
Abstract
Glass ionomer cement (GIC) is a tailor-made material that is used as a filling material in dentistry. GIC is cured by an acid-base reaction consisting of a glass filler and ionic polymers. When the glass filler and ionic polymers are mixed, ionic bonds of the material itself are formed. In addition, the extra polymer anion reacts with calcium in enamel or dentin to increase adhesion to the tooth tissue. GICs are widely used as adhesives for artificial crowns or orthodontic brackets, and are also used as tooth repair material, cavity liner, and filling materials. In this review, the current status of GIC research and development and its prospects for the future have been discussed in detail.
Introduction
Glass ionomer cement (GIC), an acid-base cement, is formed by the reaction of weak polymeric acids with inorganic glass powder [1]. GIC has multiple advantages: First, it adheres specifically to the teeth to prevent corrosion or leakage. Second, there is slow release of fluoride ion over time to maintain dental health. Third, its color is very similar to that of human teeth [2,3]. Despite the advantages of GIC, further improvement is required in terms of its mechanical characteristics. In order to improve the mechanical strength of GIC, the resin-modified glass ionomer (RMGI) was developed; it has an additional monomer compared to GIC and improved mechanical strength through photopolymerization and acid-base reaction [4,5]. RMGI obtained by resin curing has improved physical properties, but the amount of the released fluoride ion, which is important in preventing dental caries, is low [4]. Studies have reported on the manufacture of GIC using macromonomer and viscosity dilution materials to exclude the effects of water and the production of a material known as a compomer [6].
Clinically, GIC is applied close to the pulp. However, it is difficult to use RMGI in deep underlined cavities. In dental clinics, either GIC or RMGI may be used, depending on the purpose. There has been a recent focus on the study of “smart” materials that confer biocompatibility and cause remineralization, while maintaining the physical properties of materials [7]. Bioactive glass (BAG), composed of NaO, SiO, PO, and CaO, is known to be used for the loss of osseous tissue; therefore, a study was conducted to increase the biocompatibility of GIC by adding BAG to GIC [3,8]. Studies have also reported an increase in biocompatibility with the addition of synthetic hydroxyapatite (HA) to the inorganic components of GIC, since HA is highly analogous to the major components of tooth enamel or dentin in terms of structure [7].
In this review, we will describe the history of the development of GIC and determine the direction that GIC research should take in the future.
Glass ionomer cement
GIC is a combination of silicate and polycarboxylate that releases fluoride and attaches to dental tissue. It is used in a variety of applications, including the filling material of dental cervical lesions; the restoration of children’s teeth; the core construction of tubular fluid; and the adhesion of tooth fillings [9]. GIC was first introduced in 1972 by Wilson and Kent [10]. It consists of a water-soluble polyacrylic acid and fluoroaluminosilicate glass. When the silicate powder and polymeric liquid are mixed, an acid-base reaction takes place (Fig. 1). As metallic polymer salts begin to form, gelation begins, and continues until the cement hardens. Early GIC was considered an alternative to amalgam as tooth filling material. However, the mechanical properties of early GIC were not as advantageous as those of amalgam and required further improvement. Thus, the metal-reinforced GIC was first introduced in 1977. Williams et al. [11] described the addition of silver-amalgam alloy powder to GIC to increase the strength of the cement and provide radiopacity at the same time. However, both early GICs and metal-reinforced GICs had low viscosity, making them uncomfortable for clinical use. To overcome these issues, high viscosity GICs called viscous or condensable GICs were developed [12,13]. These materials were used in atraumatic restorative treatment in the early 1990s [14]. The developed materials are composed of fine glass particles and high molecular weight anhydrous polyacrylic acids and possess a high powder/liquid mixing ratio, resulting in fast setting time and conferring high viscosity [13,14]. The setting reaction mechanism of high viscosity GICs is the same as that of conventional GICs based on the acid-base reaction.
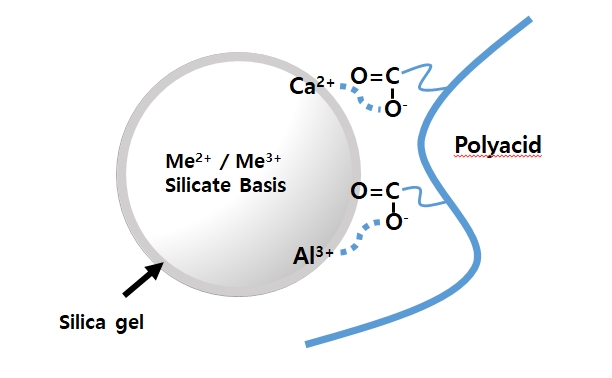
Model of ionic bond formation with inorganic filler and polyacid. When calcium fluoroaluminosilicate filler and polyacid are mixed, the carboxyl ion of polyacid is ion-bonded with aluminum and calcium ion in the silicate filler.
GICs release biologically active ions, fluoride, sodium, phosphate, and silicate that are biologically beneficial around the medium, therefore, these ions are naturally bioactive substances [15]. As more of these ions are released under acidic conditions when compared to neutral conditions, GIC can lower the pH of the surrounding medium under acidic conditions [15].
Resin-modified glass ionomer cement
RMGI is composed of resin added to the GIC. Due to resin addition, the binding strength, tensile strength, and compressive strength of the GICs are maintained and their solubility in aqueous environment is lowered, thereby improving the shortcomings of GIC [5,9]. Resin in RMGI is obtained by first putting the monomer into the liquid component of the GIC and then photo polymerization. Ultraviolet irradiation results in monomer polymerization, followed by an acid-base reaction, which improves mechanical strength (Fig. 2). Owing to this improved mechanical strength, RMGI is widely used as a dental filling material.
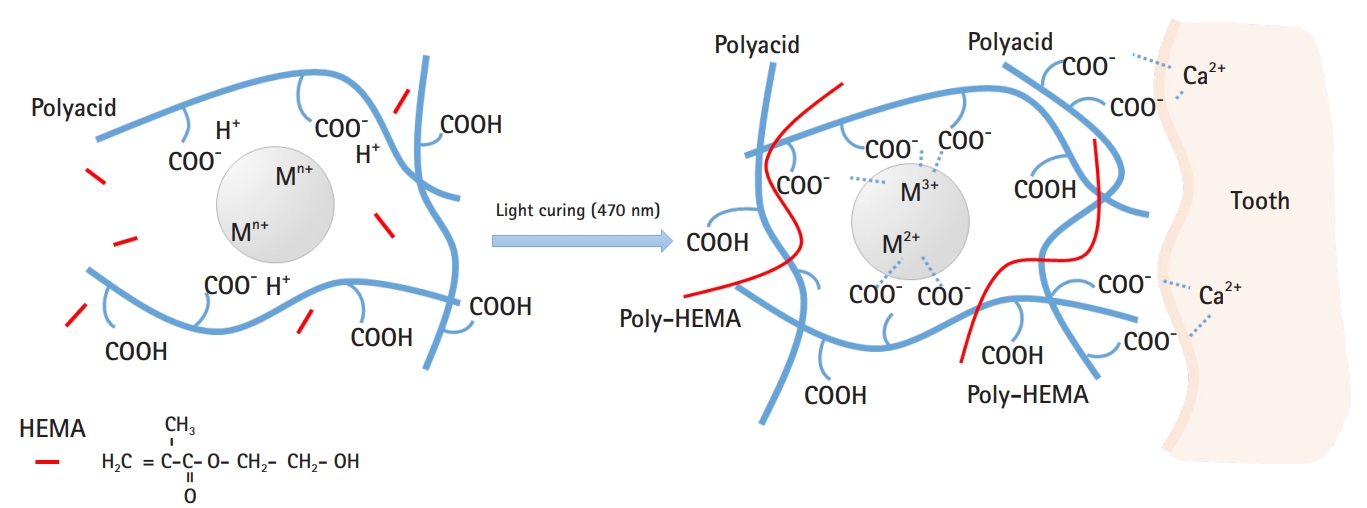
Model of interaction between resin-modified glass ionomer (RMGI) and dental tissue. When the filler, polyacid, and 2-hydroxyethyl methacrylate (HEMA) are mixed and irradiated, HEMA polymerizes and becomes poly-HEMA, acting as a bridge, followed by acid-base reactions of polyacid and filler. Meanwhile, carboxyl residues in polyacid are strongly ionized with calcium present in tooth tissue, allowing RMGI to adhere to teeth.
Earlier, Mathis and Ferracane [16] attempted to manufacture dental filling materials by mixing GIC and a composite prepared by mixing resin with commercial GIC. The resulting material did not exhibit clinically acceptable properties but it did demonstrate the possibility of combining acid-base and resin polymerization settings within a single material. RMGI, which is obtained by light curing, was developed in 1992 [5]. The basic acid-base reaction in these materials is mainly supplemented by the second resin created by light curing [5,17]. They are GICs containing a small number of monomers that can be polymerized in aqueous medium. Another method has also been reported that alters the side chain of polyalkenoic acid, but the GIC is still prepared through mechanisms based on acid-base reactions [7]. The term ‘resin-modified glass ionomer’ means that resins are formed, however, they retain the characteristics of glass ionomers [4]. With regard to the materials in the wider context of material science, RMGIs are all ‘composite materials’ as they consist of a matrix phase and a dispersed phase. The variation in the composition of commercial materials could then be considered to be continuous on a scale from purely resin-matrix produced by photo irradiation to purely salt-matrix produced by acid-base reaction [4]. One example of resin additives in RMGI is the addition of methacrylate to polyacrylic acid. In the preparation of these materials, the basic acid-base reaction is replenished by light curing. Another example of RMGI is polyacid-modified composite resins composed of macro-monomers, which are commonly used in composite resins, containing bisphenol A-glycidyl dimethacrylate (bisGMA) or urethane dimethacrylate with a small amount of acidic monomer [18,19]. They use the same ion-releasing glass as do the filler particles used in conventional GIC, however, they are small in size. The initial setting reaction is initiated by light curing, followed by an acid-base reaction after water absorption [20].
The release of fluoride from tooth filling materials is very important in terms of preventing tooth corrosion. Many researchers have reported that RMGIs can release fluoride at a rate similar to that of conventional GIC [3,20,21]. However, this release rate can be influenced not only by the formation of complex fluoride derivatives by reaction with polyacrylic acid, but also by the type and amount of the resin used for light polymerization [22-24]. Depending on the storage environment, fluoride is released from RMGI for the first 24 hours [20,25-27], then the amount of releasing fluoride decreases after 7 days, and stabilizes at 10 days to 3 weeks [20,24,28,29]. Fluoride release is affected by variables such as matrix component, filler, and fluoride content [20,30-33]. In addition, it is also affected by experimental factors such as storage environment, number and frequency of preserving solution changes, composition and pH of saliva, plaque and pellicle formation, powder-to-liquid ratio, mixing, curing time, and exposed surface [20]. Fluoride release from RMGI in artificial saliva containing esterase was proved to be higher than in artificial saliva with no enzyme [20]. Bleaching and brushing did not affect fluoride release. Removal of the outer layer of the restoration by air polishing or finishing increased fluoride release. When the surface of the restorative material was covered with an adhesive or a surface coating agent, contamination due to moisture and dehydration was prevented in the initial stage, and fluoride release was reduced by 1.4 to 4 times [20]. Mousavinasab and Meyers [34] studied the amount of fluoride released from four kinds of GIC (Fuji II LC, Fuji IX Extra, Fuji VII, and Fuji IX; GC Corporation, Tokyo, Japan), one compomer (Dyract Extra; Densply Detrey GmbH, Konstanz, Germany), and one giomer (Beautifil; Shofo Dental Corp., San Marcos, CA, USA). There was a significant difference in fluoride release depending on the type of material and time; GIC released more fluoride than the compomer and giomer. Khoroushi and Keshani [3] and Mousavinasab and Meyers [34] emphasized the role played by the amount of GIC matrix used, in releasing fluoride ion of materials.
Compared with GIC, RMGI shows improved mechanical strength but decreased biocompatibility. This is because the 2-hydroxyethyl methacrylate (HEMA) monomer escapes from RMGI mainly during the first 24 hours [2,35]. The amount of HEMA released depends on the photometric intensity of the GIC [2,35]. HEMA penetrates the dentine [2,36] and is toxic to pulp cells [2,37]. As mentioned above, the mechanical properties have been improved at the same time the working time has been reduced, but its ability to prevent cavities is relatively low owing to the low release of fluoride and its biocompatibility remains unsatisfactory because of HEMA.
Polyacid-modified composite resins (compomer)
The mechanical properties of the GIC limit its applications because it is composed of carboxylic acid groups that make the resin easily interact with water. Polyacid-modified composite resins, commonly known as compomers, are used for aesthetic materials for oral rehabilitation, especially dental caries treatment [6,38]. This material was introduced to clinical dentists in the early 1990s [6,39] and was proposed as a new dental material that combines the existing synthetic resin aesthetics with the fluoride release and adhesion capabilities of GIC [6].
The main feature of compomers is that they do not contain water and most of the components are identical to those of composite resins. Typically, these are bulky macro-monomers, such as bisGMA or its derivatives and/or urethane dimethacrylate, which are mixed with viscosity-reducing diluents, such as triethylene glycol dimethacrylate [6]. These polymer systems are filled with non-reactive inorganic powders, such as quartz or a silicate glass, such as SrAlFSiO4 [6,40]. Powders are coated with a silane, which strengthens the bond between the filler and matrix of the set material [6,41]. The compomers also contain additional monomers that are different from those of conventional composites; therefore, they contain acidic functional groups as a very minor component. The most widely used monomer of this type is TCB, which is a di-ester of 2-HEMA with butane tetracarboxylic acid [6,40]. In addition, compomers also contain reactive glass powders similar to those used in GIC [6,38].
Compomers are designed to absorb water [6,41,42], and soaking in water can lead to a 2% to 3.5% increase in their mass [41]. It has been shown that this water absorption process involves neutralization of the carboxylic acid group. Neutralization is controlled by the rate of water diffusion and is therefore a rather slow process [42]. The mechanism through which compomers absorb water to promote neutralization is found to have a negative effect on their physical properties [43,44]. This mechanism is different from that of conventional composite resins, which are known to absorb moderate amounts of water without significant alterations to their mechanical properties [44]. Adusei et al. [45] conducted the most comprehensive study of the adverse effect of water on compomers. For all tested materials, there was no difference in the measured parameters after 24-hour storage in wet or dry conditions. However, for most materials, all strength measurements tended to decrease over a 4-week period. Not all physical parameters showed reductions with long-term storage in water. In addition, it was found that microtensile strength and surface hardness appeared to remain unaffected [46,47].
The presence of minor amounts of both acid functional monomers and basic ionomer-type glass confers new properties to the material, namely, the ability to absorb moisture to trigger an acid-base reaction that can lead to the release of fluoride and creation of an acidic environment [6]. However, some studies have shown that water uptake reduces mechanical strength by up to 40% over several weeks; therefore, these clinically desirable features income at a price [44]. Conversely, clinical studies have shown that these materials perform well in a variety of applications. The decrease in mechanical strength due to water uptake does not appear to be of clinical importance, and these materials are suitable for use in vivo [48,49].
A recent study on improvements in glass ionomer cement function
Several efforts have been made to enhance the properties of GIC while maintaining the bioactivity gained by releasing the ion. However, it was necessary to develop a “smart” material that can overcome the adverse effects of the resin monomer and further induce remineralization on the defective dentin. Efforts have also recently been underway to improve physical properties and biocompatibility by using both BAG and HA as fillers.
1. Glass ionomer cement containing bioactive glass
In some recent studies [18,50-53], BAG has been used with GIC to improve bioactivity and induce tooth regeneration. The use of bioactive materials has attracted attention in dentistry, particularly for the purpose of dentin remineralization. The main inorganic component of the GIC comprises Si, Al, and Ca and is ionized with polyacid, so it does not exhibit decomposition performance [10]. Meanwhile, BAG contains specific weight percentages of Si, Na, Ca, and P and was introduced by Hench in 1969 as 45S5 Bioglass with the following chemical composition and weight percentages: 45 wt% SiO2, 24.5 wt% CaO, 24.5 wt% Na2O, and 6.0 wt% P2O5. BAGs are amorphous silicate-based materials which are compatible with the human body and can stimulate new bone growth while dissolving over time [54].
In clinical situations, BAG was first used as a biomaterial to replace the loss of osseous tissues. BAG is able to bind strongly to bone via the formation of HA and firm bonding between the collagen and HA, and the body therefore tolerates the material well [3,54]. This material was initially used in the reconstruction of bone loss due to periodontal diseases in bony defects [3,54]. BAG has recently been used in the treatment of dentinal hypersensitivity; fine BAG particles are incorporated into toothpaste or applied to tooth surfaces. BAG attaches to the dentin surface and quickly forms a hydroxycarbonapatite layer, which seals the tubules and relieves pain [3].
Some researchers have studied the physical and chemical properties to evaluate the effect of BAG materials on tooth structure. There are several studies on the effect of BAG addition on the physical properties of RMGI [3,53,55,56]. Although the compressive strength of the composition is reportedly slightly reduced, it is much higher than that of the GIC containing BAG. Yli-Urpo et al. [50] added BAG to GIC and evaluated its physical and biological properties. They reported that the experimental composition is bioactive under physiological conditions and is capable of mineralizing human dentin in vitro [3,50].
Adding BAG particles to GIC decreases compressive strength and the modulus of elasticity [50,55,57]. This suggests that the BAG particles might be only loosely attached to the GIC matrix. Thus, BAG particles probably acted as fillers that had not been adhered into the GIC matrix, leading to decreased compressive strength and modulus of elasticity [50]. Therefore, the development of bioactive GICs, that does not involve a deterioration in mechanical properties, seems to be needed. Main research has been specifically focused on the application of nanoparticles to dental materials, including GICs, to improve the mechanical properties of the matrix and strengthen communication with cells derived from dental tissue to facilitate regeneration [57-61]. Several nanomaterials such as hydroxyl- (or fluoro-) apatite, titanium oxide, zirconia, and resin and combinations thereof have been incorporated into the existing GIC. One of the nanoparticles indicated for use in GIC is a BAG nanoparticle [7,62,63]. The BAG nanoparticle, combined with the matrix of GIC, increases surface area and biological activity and greatly improves mechanical/biological properties as an additive per particle weight over that of conventional micro-sized BAG particles [64,65].
2. Glass ionomer cement containing hydroxyapatite
HA has been beneficial in the field of dentistry due to its unique radiopacity and other properties [66-68]. The application of current nano-sized biomaterials is known to be potentially more useful in dentistry. They have wide applications because of greater strength, polishability, and aesthetic value than commercial modifiers [69,70]. Recent advances in the synthesis of HA [71] in various sizes and forms have enabled HA to be used as a biocompatible filler for natural tooth materials. In addition, HA showed excellent biological activity and played an important role in orthopedics because of its bone-inducing and bioactive properties [66,72].
Nanotechnology involves the use or modification of 1 to 100-nm materials [7,73-75]. Major applications of nanotechnology in dentistry include surface modification of implants [76], enhanced polymer composites with nano-sized particles [74], and caries prevention [77]. Recent research shows that the addition of nanoparticles or nanoclusters increases the mechanical strength of tooth fillers such as resin composites [78-80]. Similar attempts have been made to improve the mechanical properties of the GIC using nanotechnology [67,81]. Introduction of nano-sized apatite not only maintains the mechanical properties of the GIC at all times, but also increases the release of fluoride ions [33,67]. Studies have also reported that GIC containing nano-sized apatite has better biocompatibility than conventional GIC [82,83]. Haider et al. [83] reported that there are differences in biological properties depending on the shape of the nanoparticles incorporated into the nanofiber scaffold. In their experiment, nanorod HA showed a better biocompatibility than spherical HA. In the HA effect study on GIC, nanorod HA-fixed silicate showed better cellular compatibility than the non-fixed silicate (Fig. 3).
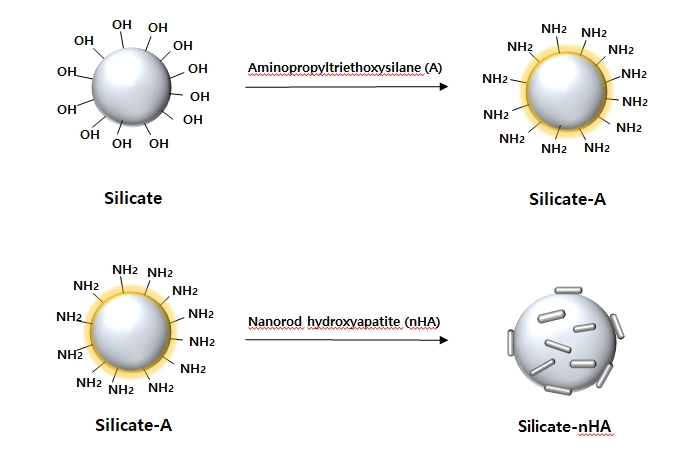
A schematic diagram that binds the nanorod hydroxyapatite (nHA) to the silicate surface. Aminopropyltriethoxysilane (A) is a coupling agent used to conjugate amino groups to the glass surface. The amino acids introduced on the surface of the silicate can react with nHA fixed carboxylic acid to produce silicate-nHA.
Apatite crystals increase the crystallinity of cured matrix, further stabilizing the hardening cement and improving the bond strength with the tooth structure [74,84,85]. Increasing fluoride release can reduce secondary caries around the restoration site [73,86]. However, the possibility of interfacial failure of glass and bioceramic can be a problem that can affect the physical properties of the cured cement [87]. The crystals of nano-HA preferentially remineralize enamel [7,88,89]. Recent reports suggest that the nano-HA-modified resin composite has improved mechanical properties over the unmodified resin composite [7,90,91]. Similarly, adding nano-HA or nano-fluoroapatite to the powder content of GIC had a positive effect on compressive, tensile, and flexural strength of the cured cement [67]. Fourier-transform infrared spectroscopy showed that adding apatite to GIC powder has been found to increase the crystallinity of cured GICs, which in turn improves chemical stability and water insolubility [67,92]. This results in a better survival rate than that observed with commercialized GICs [67].
The improved mechanical properties of GIC modified by HA are due to ionic bonds of polyacrylic acid and HA crystals [92]. As a strong ionic bond is formed between the calcium ion of the tooth structure and the crystal of the apatite of the cement, the GIC containing nano-HA is expected to strongly bond to the surface of teeth (Fig. 2) [33]. In addition, reducing the particle size of HA from a micrometer scale to a nanometer scale significantly increases the surface area, and improves infiltration into dentin and enamel pores where crystals have been demineralized; this can improve bonding at the tooth-ionomer interface [93].
HA infiltered GIC, called glass carbomer, includes substances that are established by the acid-base reaction between the aqueous polymer acid and the ion leaching base glass, but they also include substances not commonly included in glass ionomer formulations [94]. As such, the bioactive component acts as a secondary filler. According to solid state nuclear magnetic resonance spectroscopy, this filler is actually HA [95] and is included to promote the formation of enamel-like substances in contact with the tooth, as previously studied with GIC used as fissure sealants.
Since glass carbomers contain a higher proportion of glass than that in conventional GIC, as well as HA fillers, the set glass carbomers are brittle. Silicone oil is added to overcome this problem [96]. It strengthens the material and remains bound by hydrogen bonding. The setting of glass carbomer involves two parallel reactions, one involving the glass plus polyacid and the other involving HA plus polyacid. Both are acid-base reactions, resulting in an ionic crosslinking polyacid matrix containing embedded filler. However, the filler is not only ion-depleted glass, but in this case also contains a partially reactive HA. Thus, the matrix is similar to that obtained using conventional GIC, except that it contains polydimethylsiloxane oil [97].
There are only preliminary studies on the clinical use of glass carbomer thus far; however, no long-term studies have been conducted for this material. Consequently, the durability of this material in the oral cavity of patients is not yet known.
Conclusion
Since the last decade, interest in the use of “smart” bioactive materials has been growing in dentistry, especially with the aim of remineralization of dentin. More predictable treatment results can be obtained with RMGI’s superior handling characteristics, combined quality during final overlay restoration, and possibility of immediate restoration placement. Therefore, future studies should focus on these materials, especially on their cytotoxicity, quality of induced dentin bridges, and protocols for higher bonding strength during final restoration.
Currently, nanotechnology is used to develop nanoscale glass filler to enhance biocompatibility. Furthermore, various studies are being conducted to develop a material that brings high biocompatibility and mineral inducing potential by adding biocompatible nano-sized HA to RMGI. Irrespective of the clinical suitability of the material, clinicians will probably not select materials that are difficult to handle. Thus, a more biocompatible material based on RMGI need to be developed for extensive clinical use in future.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Author contributions
Conceptualization, Formal analysis, Resources, Supervision, and Validation: SK; Data curation: EYP; Writing-original draft: EYP, SK; Writing-review & editing: EYP, SK.
